|
My partner and I recently melted down bismuth using a hot plate. Once melted down, it has characteristics of meted tin, almost like silver water. It moves easily in a metal measuring cup, and hardens after some cool down time. Our goal was to dump out excess liquid bismuth around half way into the cool down time, right when the crystallization is at it's peek. Looking Back: What happened?
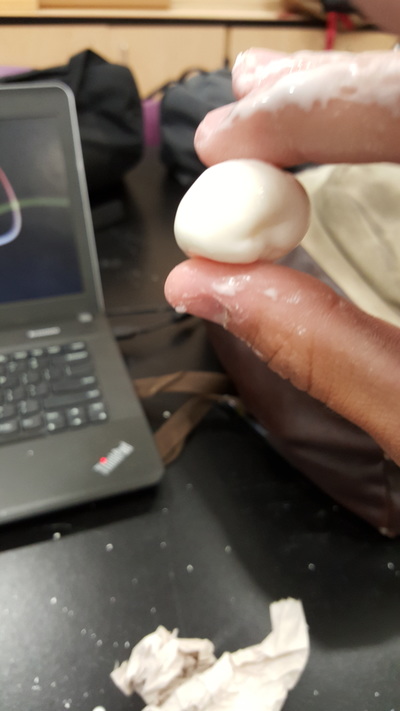
Our first few times attempting to make bismuth crystals were all busts. The crystals were not creating on their own and therefore just branching off of the sides. To make a crystal grow in the center of the container, we dropped a very small crystal of bismuth to trigger the crystallization process. this worked very well. The bottom picture shown is one of our many successful crystals of bismuth. Looking Beyond: What do you think has happened? What’s the science? Bismuth is fairly easy to understand. It submits to heat with little resistance, as it's a fairly brittle substance when thin. When it cools from a liquid state, it goes through a crystallization stage, and our goal was to dump out the rest of the bismuth once it was around halfway hardened. The science behind crystallization boggles my mind to this day, considering how intricate and precise things can be, naturally. The science behind our mini crystal triggering a bigger crystal stage is fairly simple. The temperature of the mini crystal is much colder compared to that of the surface bismuth, and when it cools is when the crystallization happens, that is why it triggers a crystallization process right at that point. Looking inward: I believe we got our desired results our final time around. We got beautiful crystals, brittle they might be. I am quite pleased with this experiment especially considering how un-orthidox the process is. I think we might have gotten bigger,better crystals if we used a metal pot to cook up a big chunk of bismuth. Looking outward: Collaboration and Impact I think my partner (Bijan S.) and I worked well together, especially because of our communication; and that is a very key quality to have in a partnership when working with dangerous materials and equipment as we were. Looking Forward: What next? If I could do this again I would use a metal pot to cook up more bismuth at one time, so that way our crystals will be bigger and theoretically more beautiful. Take aways: Bismuth makes cool jewelry
0 Comments
The Galaxy Goose lava lamp project was finished today, and all that is left is to drop a few more drops of salt water to bring the buoyancy to the perfect level. Overview: I purchased many things from home depot throughout this past week, including multiple hose clamps, electrical tape, light bulb base, different kinds of glue, and a can of tomato paste.
After gathering all of our materials, with some help from Andrew we created our blue lava using benzol alchahol and a blue sharpie ink capsule. After squeezing out the ink from the sharpie. our lava was ready to go. Next, we poured 2 part salt water after pouring 8 parts regular water. This is where the fine tuning of buoyancy comes in. While the bottle was warm, my partner (Bijan S.) and I fine tuned our lava lamp. I enjoyed this project a lot. I learned how much benzol alchahol separates itself from water and salt water, like an oil. On a molecular level, since heat expands its only natural for our "lava" to expand and rise, but since our lamp is on the bottom of our lamp, there is little to no heat on the top. Therefore the water is colder up top, cooling our lava as it rises making it fall, and repeat. One question I had during this project was what chemicals are used for comercial lava lamps that you purchase? Are they different from the chemicals we used? In my opinion, we definitely got our desired results. A very happy Galaxy Goose If I could do this project again, I would experiment with crazy bottles. Scotch bottles, bottles from distant places, etc. Today was a milestone in Bijan S. and my project because we finally finished the base to our lava lamp project. It was difficult to glue the lamp base to the bottom of our can so I designed a hose clamp system to keep it solidly in place. We were also able to put in our blue lava! After many of our classmates already experimenting they found a good mixture of salt, water, coconut oil, and lava.
(picture of lava lamp here) Tomorrow we will heat it up and hopefully get the rise and fall effect we want. Today I was able to play with dry ice and study it's properties. I have previous experience with dry ice but it was nice to experiment with it once again. The sub-zero ice-like substance was very fun to experiment with. When dry ice "melts", it skips the melting part entirely and turns to a gas. Because the air around it is much warmer (room temp), dry ice has a constant "melting" or evaporating carbon dioxide gas. When put in something warmer than room temp, it gives off even more carbon dioxide gas.
For an experiment, I put a chunk of dry ice in a balloon, and doing so made it slowly self-inflate. I then put quarters on the ice and let them freeze. It made some funny sounds. Putting dry ice in warm water was fun to see all the vapor pour out of it, but eventually it became freezing water. I have started brainstorming my DIY lava lamp, and my first ideas have changed a lot. At first, I was going to use a 500ml glass bottle of mexican coke but looking at how narrow it is I am considering a bigger bottle of a different drink. I am also gonna find my old lava lamp base for this project.
When quick pressure comes upon Oobleck, it hardens until little to no pressure is applied. When little to no pressure is being applied, it turns into a thick liquid.I am not sure if Oobleck is a liquid or a solid, if I had to guess I'd say it is a liquid. Oobleck always comes back to being a thicker liquid, for it to be considered a solid it has to be acted upon by pressure. But truthfully it is neither liquid or solid, it is called a non-newtonian fluid.
On a molecular level, Oobleck isn't far from mud. When mud is acted upon with pressure quickly, it has a similar outcome (hardens temporarily). I think some form of drag is happening, like drag on a fishing reel or when falling from the sky.There is minimal drag when minimal pressure is being applied. And lots of drag if you were to hit Oobleck or mud, creating lots of quick pressure. Picking it up and tossing it around made me feel like a young boy again. It reminded me of when I made Oobleck back in elementary. Its silly puty 2.0 to me. Its almost like I am playing with outer space matter. ''Know where the safety equipment is in the room, before your classmate meets their doom.''
Contribution Credits: Bijan S. Gabriel M. |
AuthorGabriel M. Archives
December 2015
Categories |














 RSS Feed
RSS Feed
