|
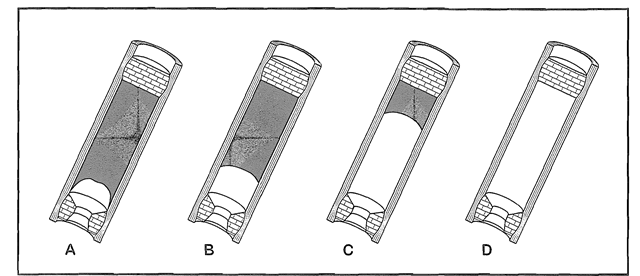
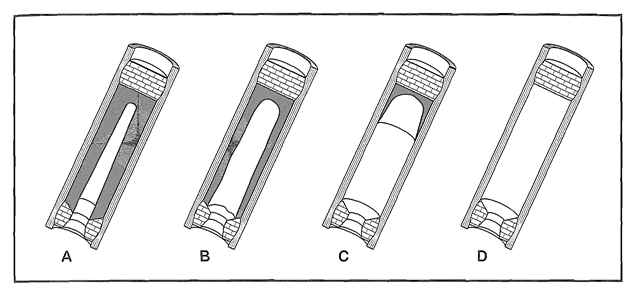
Forming a group: Phillippe and I have been friends this year, and I could tell he put a lot of work into his projects, as do I, which is why I thought we would work well together. I do not have much background on rockets, so working with Phillippe would help educate me on rockets quicker and easier. I have a strong feeling Phillippe and I will work very well together because we both have the drive for a good grade. Model Rocket Motors: There are two general types of model rocket motors, end burner motors and core burner motors. In the next section, end burner and core burner model rocket motors will be explained to my best ability. End Burner Motor: First off, an end burner engine is an engine that is known in amateur rocketry. A end burner rocket engine is an engine that has a cylindrical stick of fuel, with no gaps between the casing wall, which in this case is a round piece of cardboard, that can withstand lots of pressure. These engines have a bit of room between the fuel and the igniter, just enough so the motor can accept the igniter (in our case, an E-Match). The purpose of an igniter (or E-Match) is so that the rocket engine can be swiftly ignited from a safe distance. With our type of rocket engine, there is much more surface area for fuel to burn than an end burner, creating more pressure and ideally more thrust. These engines are used widely, and worth making if they are properly made. Some people want a small initial thrust and a long burn time after, and some want all the fuel to pretty much blow out of the bottom end. Core Burner Motor: The second motor that will be described is a core burning motor. These have a bored out hole, or also known as a core, running through the exact center of the fuel of the motor. These motors are much more dangerous than end burning motors. They are much more likely of exploding before launch due to the surface area and fuel ratio. When the E-Match is lit, the burn travels quickly all throughout the motor, giving it an intense inertia (initial thrust). There are multiple ways of coring a rocket motor. Our method was after the rocket and its fuel had been compacted using a modified press, a holder and a drill press were used to core our motors at the exact size we chose. Below and above this paragraph, are both the end burner (top) and the core burner) from before being lit and after the fuel runs dry. The Two Types Of Rocket Fuels: There are two types of fuels, and these mixtures can very. The two types of fuels are, potassium nitrate/sugar or black powder.
Potassium Nitrate/Sugar: The first rocket fuel is potassium nitrate/sugar mix. This propellant consists of 3 groups; the fuel, the oxidizer and the additive(s). In this case, the fuel is sugar. You can use all types of sugar(s) but the most used sugar is sucrose. And as an oxidizer, what is used is KNO3, which is potassium nitrate. With rocket candy the average ratio for the fuel is 65% KNO3, and 35% sugar mix. Sometimes people add 1% iron oxide, which makes the thrust rate amazingly higher. These engines can been melted down to a paste, or can be pressed with 1,000's of pounds. The reason for it needing to be packed with thousands pounds of pressure, is you cannot have any air pockets in the motor. If you do, your thrust will go down and your engine will not work that great. Black Powder: The second rocket fuel is a black powder rocket motor mixture. Black powder is usually used in model rocket motors. What these motors consist of is charcoal, sulfur and potassium nitrate. Everyone has there own ratio opinion, including us, but the most used one is 75:15:10. When people are making these engines, the amount of each component can be adjusted, which will change how the black powder burns. Most of the time these black powder rocket motors are only produced in small sizes, due to these being extremely explosive, ESPECIALLY when cored.
0 Comments
Today, we took a closer look at combustion reactions. This was important to look into because our big project for this chemistry section is on making rocket motors from scratch; also known as a combustion reaction. Here's what we did: First, we took a look at the fundamentals of atoms and the things that would be involved. Next, Andrew took multiple volumetric flasks filled 30% of water an mixed in a powdered form of citric acid. Then, we took small strips magnesium and coiled them. After dropping the pieces in, we quickly put a small balloon over the top of the flask. What I took away from this was how atoms react to different things. Up until now, I did not know any of the concepts behind atoms and what they do when meeting other atoms with the same and different energy levels and electron count.
1)
a. I chose a time lapse of a beach sunset because it is something that happens every single night, and at times goes un-noticed. There is beauty and science to every sunset. What most people don't usually think about is how the sun isn't actually going to sleep, rather stays on 24/7 as WE circle IT. b. I used the internet to further investigate the track that our earth takes for sunsets to happen at all. c. n/a d. I thought about how even though the sun and clouds are 92 million miles away from each other, the sunset we immediately can see with our eyes looks like they're side by side. e. I wonder if the planets we aren't able to travel to see our sun? Or possibly to sustain life, have their own sun? Is there some kind of probe we can send out that can sense another sun, or find presence of another one? 2) a. I learned that the sun is much bigger than I had originally thought, something to think about when I watch the sunset of the days to come. b. Almost everyone knows that our planets circle the sun. So, what makes the sun so special? How is it self sustaining since the beginning? Could we make our own sun on a small scale, that is self sustaining, to create infinite power? Will we advance far enough to do so before we run out of resources to live? How about something simpler: Are there planet(s) hiding behind the sun in a constant orbit that makes it to where we never see it? No, in fact if there were planets that we didn't know about, we would inevitably orbit close to that planet. And as far as our self sustaining sun goes, what we do know is that there are two forces at work; gravitational collapse and thermal expansion. Gravitational collapse requires no energy, and would reduce the sun to about the size of the earth if unchecked. But as the sun contracts, it heats up enough for nuclear fusion to start, producing even more heat. Heat causes the sun to expand, counterbalancing the gravitational force that causes it to contract. The result is a self-sustained balance between contraction and expansion. The more it collapses, the more heat is generated to make it expand. The more it expands, the less heat is generated, allowing it to collapse again. c. If I were to continue research on the sun, I would want to talk to a genius on the subject, since you can't really trust anything on the internet (INCLUDING THIS POST). d. My main take away from this would be to work on not taking sunsets for granite, not seeing it as something visually pleasing but something incredible happening million miles away. Today was launch day for our rocket. My partners were Bijan S. and Trent. Our goal was to build a working model rocket that after launch, would come down with it's own parachute. Build Notes: To build our model rocket we had to first assemble the engine mount. To build the engine mount, we had to hold different pieces together for the gorilla glue to dry. Once fully hardened over night, we were able to move on to the next step of assembling the fins. This was more difficult than the motor mount because the fins had much less surface area for the glue to grab. After some trial and error we were able to mount all three fins on very solid. Final step was to make the parachute device. We did this by tying the parachute to the nose piece, and then a thick rubber band from the nose piece to the body of the rocket. Now that our rocket was built, we were able to pack our parachute and rubber band into the rocket itself. Trent then went to Fiesta island with some other students launching rockets and launched our model rocket.
Performance Notes: I think our rocket performed very well. We got our desired results and learned lots of useful information on rocketry and the chemistry behind it. When the motor is launched, it spits off power to the rocket, and since the rocket is guided by the launch pad, the rocket has more of a tendency to go straight up. And as shown in the video, ours did just that. Once it started to descend, the nose piece comes off and lets the parachute open up instantly, safely gliding it down back to earth. Today Andrew took multiple substances, took a small portion of it and added a few drops of water, and then put it over a bunsen burner. A bunsen burner is a type of propane burner that emits a calm yet incredibly hot flame over the surface area of a half dollar. Our setup had a sturdy stand holding both the bunsen burner and propane fuel can in place. The propane had to be at an an angle because of propane not being all gas. Then, a gas line was run from the propane can to the burner. We were then ready to light some stuff on fire in the name of chemistry!
Copper (Cu) = Green Sodium (Na) = Yellow orange Boron (B) = Light Green Strontium (Sr) = Dark orange Pottasium (K) = Violet Magnisium (Mg) = White (Above are results from taking multiple substances in small quantities and burning them) Looking Back: I found that although we were burning different chemicals, some tended to burn near the same color, which could probably be a topic for research. "Why do some chemicals, although different, burn the same color?" Looking Beyond: What do you think has happened? What’s the science? After a brief talk on the basics of whats happening on a molecular level, I understood that our substances are heated up to a very specific level as a result of a very specific heat being applied. When that energy is released, or the substance has it's heat source taken away, it falls back down the levels and on the way down releases light or color. Looking inward: Humans doing science - Lab Practice I think we could have gotten better results. Some particles from different substances found their way onto the bunson burner, making the chemical reaction for the other substances tainted. We were still able to see the other colors that each substance produced, even though another substance was still in play. I think we could have avoided this by having multiple metal rods to put the different substances in so that there was no contamination from the previous substances. Looking outward: Collaboration and Impact Not applicable, Andrew had most of the fun. A few students were able to come up and hold substances over the flame, their reactions were funny. Looking Forward: What next? I could definitely see myself doing a lab like this one, and I would probably use a wide array of chemicals and substances to maximize the results I get. Take aways: I was able to learn that on a molecular level, I am able to much better understand how things are reacting and why chemicals do what they do when being heated. |
AuthorGabriel M. Archives
December 2015
Categories |







 RSS Feed
RSS Feed
